Statistical Bulletin 2017 — drug-related infectious diseases
Archived content
Drug-related infectious diseases such as HIV and hepatitis B and C are among the most serious health consequences of drug use. Even in countries where HIV prevalence in injecting drug users (IDUs) is low, other infectious diseases, such as hepatitis B/C, sexually transmitted diseases, TB, anthrax, tetanus, botulism, hepatitis A, HTLV may disproportionately affect drug users.
Data tables
Use the menu below to find data sets of interest.
Graphics
Click on a graphic from the thumbnails below
Methods and definitions used
Drug-related infectious diseases such as HIV and hepatitis B and C are among the most serious health consequences of drug use. Even in countries where HIV prevalence in injecting drug users (IDUs) is low, other infectious diseases, such as hepatitis B/C, sexually transmitted diseases, TB, anthrax, tetanus, botulism, hepatitis A, HTLV may disproportionately affect drug users.
IDUs are the target group for measuring prevalence of drug-related infections. They are defined as any person who has ever in their lifetime injected a drug for non-medical purposes. In practice, almost all data on IDUs collected by the EMCDDA relate to ‘ever injectors’ among active drug users who are in contact with drug services, although some studies report data for ‘recent injectors’ (e.g. those injected in the last 12 months) among active drug users in contact with services.
The EMCDDA is systematically monitoring HIV and hepatitis B and C among injecting drug users (prevalence of antibodies, or other specific markers in the case of hepatitis B). This is as a complement to existing notification and case-reporting systems that follow trends in the notified or reported number of cases. National notification data of hepatitis B/C are often unreliable due to under-diagnosis (large proportions of new infections are asymptomatic), under-reporting and misclassification of injecting risk. HIV case reporting data are more likely to be of better quality, although they are subject to similar problems. Other infections may in the future be added to the EMCDDA monitoring system (e.g. other sexually transmitted infections and tuberculosis among drug users), while the EMCDDA collaborates with the European Early Warning and Response System (EWRS — coordinated by the ECDC) regarding outbreaks of serious infections related to (injecting) drug use, such as HIV, anthrax, tetanus and wound botulism.
To improve HIV and hepatitis B/C monitoring in IDUs, the EMCDDA follows two lines of work:
- Collecting existing prevalence data (HIV and hepatitis B/C) and notification data (currently, hepatitis B/C notifications; HIV case reports are obtained from ECDC–WHO/Europe) in aggregate format, using a standard data reporting form (‘Standard Table 9’, since 2008 through an online data collection system ‘FONTE’ ).
- Stimulating new sero-behavioural studies in injecting drug users and stimulating increased screening of IDUs and data collection in routine settings such as drug treatment, by maintaining an expert network to discuss methods and work towards common protocols.
The EMCDDA has developed draft guidelines for the national focal points to collect the existing prevalence and notification data and a draft protocol for primary data collection through sero-prevalence studies. This draft protocol is currently in the process of revision and being transformed into a modular ‘toolkit’. Please contact the EMCDDA in case you need the latest formats and proposals for this toolkit. For more information on the current tools and methods (but not the latest revision proposals), see the DRID key indicator page.
To further improve the comparability of prevalence data in IDUs, data are collected and reported on prevalence of HIV and hepatitis in young IDUs (under 25 years of age) and new IDUs (who have injected for less than two years). These indicators, and especially the HCV data for new IDUs, may be more sensitive to changes in incidence than in prevalence in all IDUs.
In practice, the target group differs slightly between settings: sero-prevalence data from needle exchanges by definition refer to active injectors, while data from hepatitis notifications or public health laboratories may be partly based on former injectors, so additional methodological data such as service setting are also collected.
Following discussions during the annual meetings of the EU expert network, a new template was included in ‘Standard table 9’ in order to collect information on key behavioural characteristics of the IDUs in the studies’ samples. Main items include HIV testing and risk behaviour (e.g. needles or paraphernalia sharing) and other variables related to the risk of contracting a blood-borne infectious disease, e.g. homelessness or sex work. Please contact the EMCDDA for the revised formats of these items.
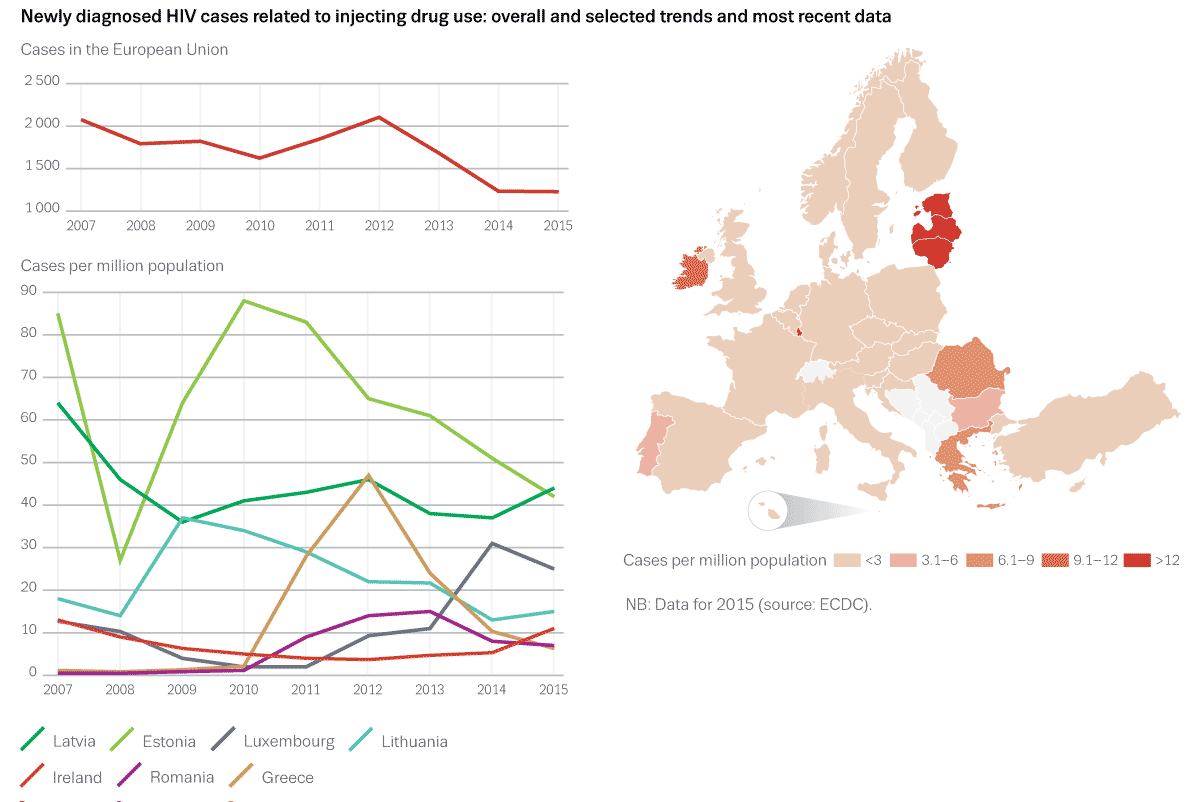
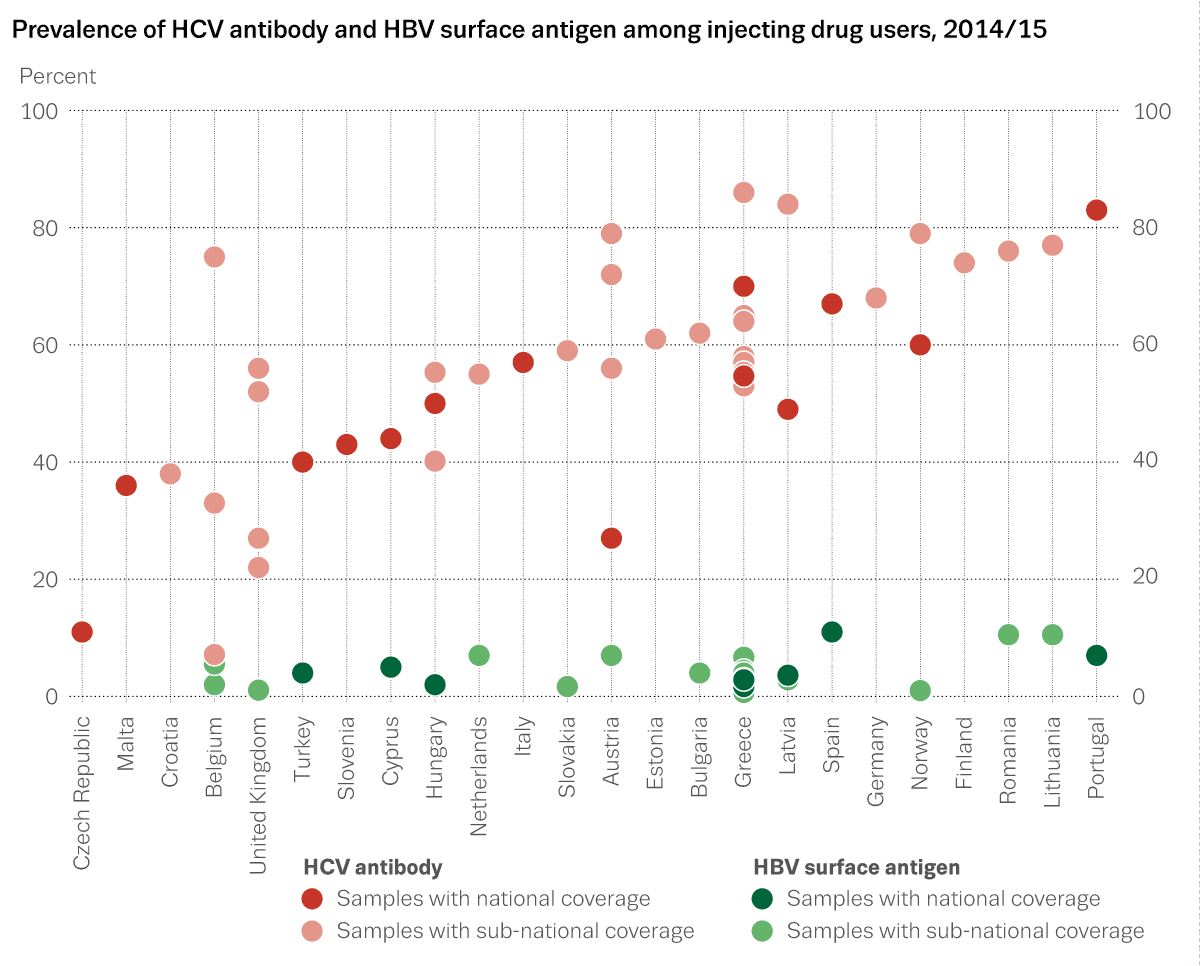
The aggregate prevalence data collection through the standard reporting form has been successful. The data provide a general overview of HIV and hepatitis B/C prevalence among IDUs in all EU Member States going back to 1996 and, in part, even before. Many countries are able to provide up to date data with national coverage and in many cases there is regional breakdown or data from key regions or cities, often unpublished and recent. These data have proven useful to provide a broad overview of the situation, including regional variation in levels and trends. Although in general they show a relatively stable prevalence of HIV and hepatitis among IDUs, they have served to document increases in HIV or hepatitis among subgroups of IDUs in some countries, including countries which are recently experiencing serious HIV outbreaks among IDUs (Greece, Romania).
However, the data are subject to important limitations: the use of varying source-types/settings (drug treatment, low-threshold, prisons, etc.) that may result in different biases, in some cases non-adherence to the basic case definition of ‘ever-IDUs’ that by inclusion of non-IDUs may lead to potentially serious downward bias, and other problems. Improving data quality and comparability proves difficult, as this depends on influencing often well-established data producing systems. Also, to get quality information on trends over time from routine diagnostic data (as opposed to well-defined prevalence studies), it is necessary to understand selection procedures for being tested and, if possible, to work towards more standardisation in the criteria for screening of IDUs in contact with services.
Finally, in response to the EU Drugs Action Plan 2005–08, the EMCDDA has set up the ‘European study group for mathematical modelling and epidemiological analysis of drug-related infectious diseases’. The group aims to use modelling techniques and data from available epidemiological studies to understand the factors (including interventions) contributing to the large differences observed in the spread of HIV and hepatitis among IDUs in Europe. After an initial phase funded by EMCDDA 2007-2008 the study group has been working with funding provided by WHO/Europe and the government of the Netherlands between 2008 and 2010. Currently the group is stimulating new analyses using data from the study group.
References
- Hahné SJ, Veldhuijzen IK, Wiessing L, Lim TA, Salminen M, Laar M. Infection with hepatitis B and C virus in Europe: a systematic review of prevalence and cost-effectiveness of screening. BMC Infect Dis. 2013 Apr 18;13(1):181.
- Vickerman P; Martin NM, Roy A, Beattie T, EMCDDA collaborative group, Des Jarlais D, Strathdee S; Wiessing L; Hickman M. Is the HCV-HIV co-infection prevalence amongst injecting drug users a marker for the level of sexual and injection related HIV transmission? Drug and Alcohol Dependence 2013 (in press)
- Mathers BM, Degenhardt L, Bucello C, Lemon J, Wiessing L, Hickman M. Mortality among people who inject drugs: a systematic review and meta-analysis WHO Bull. 2013 ; 91 (2) : 81-156. doi: 10.2471/BLT.12.108282
- Rondy M, Wiessing L, Hutchinson SJ, Matheï C, Mathis F, Mravcik V, Norden L, Rosinska M, Scutelniciuc O, Suligoi B, Vallejo F, van Veen M, Kretzschmar M. Hepatitis C prevalence in injecting drug users in Europe, 1990-2007: impact of study recruitment setting. Epidemiol Infect. 2012 ; 17:1-10.
- Sutton AJ, House T, Hope VD, Ncube F, Wiessing L, Kretzschmar M. Modelling HIV in the injecting drug user population and the male homosexual population in a developed country context. Epidemics 2012 ;4 :48-56.
- Hope VD, Palmateer N, Wiessing L, Marongiu A, White J, Ncube F, Goldberg D. A decade of spore-forming bacterial infections among European injecting drug users: pronounced regional variation. Am J Public Health 2011 Nov 17.
- Wiessing L, Likatavicius G, Hedrich D, Guarita B, van de Laar MJ, Vicente J. Trends in HIV and hepatitis C virus infections among injecting drug users in Europe, 2005 to 2010. Euro Surveill. 2011;16(48):pii=20031.
- Pharris A, Wiessing L, Sfetcu O, Hedrich D, Botescu A, Fotiou A, Nikolopoulos GK, Malliori M, Salminen M, Suk JE, Griffiths P, van de Laar MJ. Human immunodeficiency virus in injecting drug users in Europe following a reported increase of cases in Greece and Romania, 2011. Euro Surveill. 2011;16(48):pii=20032.
- Castro-Sanchez A, Shkedy Z, Hens N, Aerts M, Geskus R, Prins M, Wiessing L, Kretzschmar M. Estimating the force of infection for HCV in injecting drug users using interval-censored data. Epidemiol Infect. 2011 Sep 12:1-11.
- Hatzakis A, Wait S, Bruix J, Buti M, Carballo M, Cavaleri M, Colombo M, Delarocque-Astagneau E, Dusheiko G, Esmat G, Esteban R, Goldberg D, Gore C, Lok ASF, Manns M, Marcellin P, Papatheodoridis G, Peterle A, Prati D, Piorkowsky N, Rizzetto M, Roudot-Thoraval F, Soriano V, Thomas HC, Thursz M, Valla D, van Damme P, Veldhuijzen IK, Wedemeyer H, Wiessing L, Zanetti AR and Janssen HLA. Special Issue: The state of hepatitis B and C in Europe: report from the hepatitis B and C summit conference. J Viral Hepatitis 2011; 18, Issue Supplement s1: 1-16.
- Del Fava E, Shkedy Z, Hens N, Aerts M, Suligoi B, Camoni L, Vallejo F, Wiessing L and Kretzschmar M. 2011. “Joint Modeling of HCV and HIV Co-Infection among Injecting Drug Users in Italy and Spain Using Individual Cross-Sectional Data.” Statistical Communications in Infectious Diseases: Vol. 3: Iss. 1, Article 3. DOI: 10.2202/1948-4690.1010.
- Del Fava E, Kasim A, Usman M, Shkedy Z, Hens N, Aerts M, Bollaerts K, Scalia Tomba G, Vickerman P, Sutton AJ, Wiessing L, Kretzschmar M. Joint Modeling of HCV and HIV Infections among Injecting Drug Users in Italy Using Repeated Cross-Sectional Prevalence Data. Statistical Communications in Infectious Diseases: Vol. 3: Iss. 1, Article 1. DOI: 10.2202/1948-4690.1009
- Wiessing L, Blystad H. EMCDDA publishes guidelines on testing for HIV, viral hepatitis and other infections in injecting drug users. Euro Surveill. 2010;15(48):pii=19735.
- Wiessing L, Klempová D, Hedrich D, Montanari L, Gyarmathy VA. Injecting drug use in Europe: stable or declining. Euro Surveill. 2010;15(26):pii=19604.
- Degenhardt L, Mathers B, Guarinieri M, Panda S, Phillips B, Strathdee SA, Tyndall M, Wiessing L, Wodak A, Howard J, the Reference Group to the United Nations on HIV and injecting drug use. Meth/amphetamine use and associated HIV: Implications for global policy and public health. Int J Drug Policy. 2010 Jan 29. [Epub ahead of print]
- Mathers BM, Degenhardt L, Ali H, Wiessing L, Hickman M, Mattick RP, Myers B, Ambekar A, Strathdee SA; for the 2009 Reference Group to the UN on HIV and Injecting Drug Use. HIV prevention, treatment, and care services for people who inject drugs: A systematic review of global, regional, and national coverage. Lancet. 2010 Mar 20;375(9719):1014-1028. Epub 2010 Feb 26.
- Kolarić B, Štajduhar D, Gajnik D, Rukavina T, Wiessing L. Seroprevalence of blood-borne infections and population sizes estimates in a population of injecting drug users in Croatia. Central European Journal of Public Health. Cent Eur J Public Health 2010; 18 (2): 104–109.
- Vickerman P, Hickman M, May M, Kretzschmar M, Wiessing L. Can HCV prevalence be used as a measure of injection-related HIV-risk in populations of injecting drug users? An ecological analysis. Addiction 2010 Feb;105(2):311-8. Epub 2009 Nov 16.
- Blystad H, Wiessing L. Guidelines for Testing HIV, Viral Hepatitis and other Infections in Injecting Drug Users. Lisbon, EMCDDA, 2010.
- Wiessing L, Olszewski D, Klempová D, Vicente J, Griffiths P. EMCDDA Annual report 2009: cocaine and heroin maintain firm hold on Europe’s drug scene. Euro Surveill. 2009;14(46):pii=19410.
- Wiessing L, Likatavičius G, Klempová D, Hedrich D, Nardone A, Griffiths P. Associations Between Availability and Coverage of HIV-Prevention Measures and Subsequent Incidence of Diagnosed HIV Infection Among Injection Drug Users. Am J Public Health 2009;99(6):1049-52.
- Gyarmathy, V.A., Giraudon, I., Hedrich, D., Montanari, L., Guarita, B., Wiessing, L. (2009), ‘Drug use and pregnancy — challenges for public health’, Eurosurveillance 14(9), pii=19142.
- Wiessing, L., Guarita, B., Giraudon, I., Brummer-Korvenkontio, H., Salminen, M., Cowan, S.A. (2008), ‘European monitoring of notifications of hepatitis C virus infection in the general population and among injecting drug users (IDUs) — the need to improve quality and comparability’, Eurosurveillance 13(21):pii=18884.
- Mathers, B., Degenhardt, L., Phillips, B., Wiessing, L., Hickman, M., Strathdee, S.A., Wodak, A., Panda, S., Tyndall, M., Toufik, A., Mattick, R.P. and the Reference Group to the United Nations on HIV and Injecting Drug Use (2008), ‘The global epidemiology of injecting drug use and HIV among people who inject drugs: a review’, Lancet 372 (9651), pp. 1733–1745. Epub 23 September, 2008.
- Kretzschmar, M., Wiessing, L. (2008), ‘New challenges for mathematical and statistical modeling of HIV and hepatitis C virus in injecting drug users’, AIDS 22, pp. 1–10.
- Sutton, A.J., Hope, V.D., Ncube, F., Matheï, C., Mravcik, V., Sebakova, H., Vallejo, F., Suligoi, B., Wiessing, L., Kretzschmar, M. (2008), ‘A comparison between the force of infection estimates for blood-borne viruses in injecting drug user populations across the European Union — A modelling study’, Journal of Viral Hepatitis 15(11), pp. 809–816. Epub 28 August, 2008.
- Hedrich, D., Pirona, A., Wiessing, L. (2008), ‘From margin to mainstream: the evolution of harm reduction responses to problem drug use in Europe’, Drugs, Education and Prevention Policy 15(6), pp. 503–517.
- Wiessing, L., van de Laar, M.J., Donoghoe, M.C., Guarita, B., Klempová, D., Griffiths, P. (2008), ‘HIV among injecting drug users in Europe: increasing trends in the East’, Eurosurveillance, 13(50), pii=19067.
- Kretzschmar, M., Wiessing, L. (eds.) (2007), ‘Coordination of a working group to develop mathematical and statistical models and analyses of protective factors for HIV infection among injecting drug users’, European Monitoring Centre for Drugs and Drug Addiction (EMCDDA), Lisbon.
- Backmund, M., Reimers, K., Reimer, J., Krausz, M., Zachoval, R., Gölz, J., Klempova, D., Wiessing, L. (2007), Protective factors for HIV infection in IDUs — EMCDDA literature review, European Monitoring Centre for Drugs and Drug Addiction (EMCDDA), Lisbon.
- Reintjes, R., Wiessing, L. (2007), ‘Second generation HIV surveillance and injecting drug use: uncovering the epidemiological ice-berg’, International Journal of Public Health, 52(3), pp. 166–72.
- Vicente, J., Wiessing, L. (2007), ‘European Monitoring Centre for Drugs and Drug Addiction Annual report 2007: positive assessment of HIV in IDUs through hepatitis C still very high’, Eurosurveillance,12(11), E071122.6.
- Kretzschmar, M., Wiessing, L. (eds.) (2007), ‘Coordination of a working group to develop mathematical and statistical models and analyses of protective factors for HIV infection among injecting drug users’, European Monitoring Centre for Drugs and Drug Addiction (EMCDDA), Lisbon.
- Wiessing, L., Nardone, A. (2006), ‘Ongoing HIV and viral hepatitis infections in IDUs across the EU, 2001–2005’, Eurosurveillance 11(11); 23 November, 2006.
- Kontogeorgiou, K., Terzidou, M., Klempova, D., Wiessing, L. (eds.) (2006), ‘Protocol for the implementation of the EMCDDA key indicator drug-related infectious diseases (DRID)’, draft version 6 October 2006, Project CT.04.P1.337, European Monitoring Centre for Drugs and Drug Addiction (EMCDDA), Lisbon.
- Reimer, J., Schulte, B., Castells, X., Schafer, I., Polywka, S., Hedrich, D., Wiessing, L., Haasen, C., Backmund, M., Krausz, M. (2005), ‘Guidelines for the treatment of hepatitis C virus infection in injection drug users: status quo in the European Union countries’, Clinical Infectious Diseases 40, Supplement 5, S373–8.
- Wiessing, L., Ncube, F., Hedrich, D., Griffiths, P., Hope, V. et al. (for the EMCDDA expert network on drug-related infectious diseases) (2004), ‘Surveillance of infectious diseases in IDUs across the EU: information from the EU expert network’, Eurosurveillance Weekly 8: 040122.
- Hope, V., Ncube, F., de Souza, L., Gill, N., Ramsay, M., Goldberg, D., Thomas, D., Smyth, B., Wiessing, L. (2004), ‘Shooting up: infections in injecting drug users in the United Kingdom, 2002’, Eurosurveillance Weekly 8: 040122.
- Wiessing L. The access of injecting drug users to hepatitis C treatment is low and should be improved. Eurosurveillance Weekly 2001; 5: 010802.